Weighing up COVID-19 vaccines
16 February 2021 | Story Carla Bernardo. Read time >10 min.
By now you would have heard the disappointing news about the Oxford/AstraZeneca COVID-19 vaccine and the South African government’s decision to pause its roll-out. You might also have heard about other vaccines such as Russia’s Sputnik V or that of Johnson & Johnson. And you may be wondering about their efficacy or want to know the benefits of being vaccinated. The University of Cape Town’s (UCT) Professor Heather Zar looks at the different COVID-19 vaccines and answers some of the public’s most pressing questions.
In a recent lecture hosted by UCT’s Department of Psychiatry and Mental Health’s social responsiveness committee during a UCT paediatric refresher course, Professor Zar unpacked these issues and weighed up the various COVID-19 vaccines in terms of efficacy, safety and potential use in South Africa. Zar is the chairperson of the Department of Paediatrics and Child Health and the director of the South African Medical Research Council (SAMRC) Unit on Child and Adolescent Health at UCT.
She presented a snapshot of COVID-19 in South Africa. As of 8 February 2021, there were around 1.5 million cases and nearly 47 000 known deaths directly attributed to the virus. The median age of people infected in the second wave is 40 years and the mortality rate is among the highest in the world.
It is also now known that COVID-19 does not only cause acute illness but also chronic morbidity – what has become known as “long COVID”. The emergence of the new 501Y.V2 virus variant, which was first identified in South Africa and is 50% more transmissible, means that anyone in this country who has COVID-19 should be considered to have the variant strain.
“Vaccines are going to be a key intervention in terms of preventing further infection, disease and ultimately getting the pandemic under control.”
In terms of healthcare workers and protection from SARS-CoV-2 infection, Zar referred to a study that was conducted at the Red Cross War Memorial Children’s Hospital during the first wave. Study results showed that the prevalence of antibodies to the virus was only 10% in healthcare workers who volunteered in the study – this remained relatively unchanged three months later. This indicates that many healthcare workers remain extremely vulnerable to infection.
Thus, said Zar, “Vaccines are going to be a key intervention in terms of preventing further infection, disease and ultimately getting the pandemic under control.”
Vaccines overview
She provided overviews of the four different groups of vaccines that have been produced: messenger RNA (mRNA) vaccines (Pfizer and Moderna), adenovirus vectored vaccines (AstraZeneca, Sputnik V and Johnson & Johnson), protein vaccines (Novavax) and attenuated or inactivated SARS-CoV-2 virus vaccines (China’s Sinovac or Sinopharm). Her focus, however, was on the mRNA and adenovirus vectored vaccines.
Almost all vaccines use some component related to SARS-CoV-2’s spike protein to elicit immunity to the virus. Its spike protein enables SARS-CoV-2 to bind to and enter human cells, leading to COVID-19. By developing antibodies and immunity to the spike protein following vaccination, an individual will be protected against COVID-19 should they get infected with SARS-CoV-2.
In brief, mRNA vaccines have good published data, namely for Phase 2 (studies involving hundreds of people and which primarily assess how safe the vaccine is, different doses and immune responses) and Phase 3 (studies of thousands of people that provide information on the efficacy and safety of vaccines) trials. They have been approved for use in several countries. These vaccines are fairly unstable, however, and therefore require storage in freezers with temperatures of -70°C.
Adenovirus vectored vaccines require refrigeration and almost all require two doses, an exception being Johnson & Johnson’s product. Regarding Phase 3 results, AstraZeneca’s and Sputnik V’s have been published, while Johnson & Johnson has provided headline data.

Interestingly, adenovirus vectors have a long history of testing for vaccines against Ebola, respiratory syncytial virus Zika, and HIV.
“It is an extraordinary feat of science, but … a lot of [that] science has built on decades of work.”
“People ask how, less than 11 months into the pandemic, we have vaccines. And it is an extraordinary feat of science, but … a lot of [that] science has built on decades of work,” said Zar.
“It’s the concerted efforts of many groups around the world doing incredible work to rapidly develop and test vaccines … but it’s also the preceding decades of research that have enabled this.”
Immune responses
Immune responses are important in evaluating whether vaccines will be effective, including against variant strains. The mRNA vaccines have proven “highly immunogenic”, showing big increases in antibody responses after vaccination – both in younger people and the elderly – as well as T-cell responses, which are important for killing the virus inside cells.
The AstraZeneca vaccine achieves antibody levels between days 35 and 42 that are comparable to those in people who have had COVID-19.
“Studies have shown that antibodies to the SARS-CoV-2 spike protein last almost 465 days in most people.”
As for the duration of immunity, studies have shown that antibodies to the SARS-CoV-2 spike protein last almost 465 days in most people. However, other antibodies, such as the antibodies to the nuclear protein, last for about 60 days.
More good news is that six to eight months after people had COVID-19, there was still substantial immune memory, which means that if they encountered the virus again, there would be a robust immune response.
Clinical efficacy
Zar also provided details on the clinical efficacy of the vaccines for clinical disease (illness with symptoms – this ranges from mild illness like a cold to more severe illness like pneumonia), severe COVID-19, clinical disease due to the variant virus, and for severe disease due to the variant virus.
In summary, the Pfizer and Moderna vaccines both have very high efficacy for clinical disease and severe COVID-19 for the original virus. And while available results precede the variants, a recent study shows that antibodies effectively neutralise variant viruses, with a very mild reduction in neutralisation against the 501Y.V2 variant, further suggesting that the vaccine will be effective against the virus in South Africa.
Before the variants arose, the AstraZeneca vaccine reported 67% efficacy for clinical disease and “very good protection” for severe COVID-19. However, recently reported headline results found that it had only 22% protection for mild or moderate disease with the 501Y.V2 variant.
Importantly, these results come from a study in which participants were at very low risk of severe disease (median age 31 years, all HIV negative), and in which two doses of vaccine were given four weeks apart (a longer interval has been shown to be more effective in United Kingdom trials), so the protective efficacy for severe disease is unknown.
This vaccine is on hold for roll-out in South Africa; however, the World Health Organization has advised that it should still be used in areas where the variant virus is circulating, as it is likely to be protective against severe disease.

The Sputnik V vaccine was also highly effective against the non-variant virus. Tested in 25 health facilities in Moscow, 25% of patients had comorbid conditions, 35% were older than 50 years, and after two doses there was an overall efficacy of 92% and 100% against severe disease.
“This looks like a really promising vaccine,” said Zar, adding that the Sputnik V and AstraZeneca research teams are now working together.
Finally, there is a headline result for the Johnson & Johnson vaccine of 57% protection against clinical disease from the variant and 85% against severe disease in South Africa, but more detailed results need to be seen. This large study was done in the United States (US), South America and South Africa, with a reported vaccine efficacy of 72% in the US, 66% overall and 57% in South Africa (in the context of the variant virus).
The trial had good representation in terms of comorbidity (41% of participants had comorbid conditions) and around a third were over 60 years old. Further, most cases of COVID-19 in South Africa were due to the variant strains.
Benefits of vaccination
Zar listed a few reasons why people should get vaccinated. The first is to protect yourself against COVID-19, to reduce your chances of getting severe disease and, particularly for healthcare workers, to reduce the stress and anxiety of being in environments where there is a high potential exposure to the disease.
“I think vaccination is safe and effective, particularly against severe disease. There is a lower efficacy for the variants, but good protection for severe disease.”
Then there is the public health benefit of protecting those around you, including your household members. There is also the economic benefit: “If we can develop herd immunity, we can return to normal living, schooling, an economy that’s opened, and to travel,” said Zar, before sharing some final thoughts.
“I think vaccination is safe and effective, particularly against severe disease. There is a lower efficacy for the variants, but [there’s likely] good protection for severe disease,” she said.
According to available results at the time, Zar believes the Pfizer vaccine is a good option against the variant, and while more data is needed on the Johnson & Johnson vaccine, the headlines look promising.
The AstraZeneca vaccine may still be an option for those at risk of severe disease, as it is likely that they will be protected. Other vaccines, such as Sputnik V or Moderna, which have shown such high efficacy for non-variant strains, should also be considered as possible options.
Importantly, there must be rapid vaccination of healthcare workers, followed by vulnerable groups and, Zar emphasised, we must continue wearing masks, social distancing and washing hands to control transmission.
“We must [urge] our Department of Health to immediately roll out the Pfizer vaccine [and] potentially the Johnson & Johnson vaccine for healthcare workers, followed by those who are most vulnerable. The use of the AstraZeneca vaccine for those at high risk of severe disease should be considered.
“We need whatever vaccine we can get to protect all of us, and particularly our healthcare workers at the coalface.”
 This work is licensed under a Creative Commons Attribution-NoDerivatives 4.0 International License.
This work is licensed under a Creative Commons Attribution-NoDerivatives 4.0 International License.
Please view the republishing articles page for more information.
UCT’s response to COVID-19 in 2021
COVID-19 is a global pandemic that caused President Cyril Ramaphosa to declare a national disaster in South Africa on 15 March 2020 and to implement a national lockdown from 26 March 2020.
UCT is taking the threat of infection in our university community extremely seriously, and this page will be updated regularly with the latest COVID-19 information. Please note that the information on this page is subject to change depending on current lockdown regulations.

Global Citizen Asks: Are COVID-19 Vaccines Safe & Effective?
UCT’s Institute of Infectious Disease and Molecular Medicine (IDM) collaborated with Global Citizen, speaking to trusted experts to dispel vaccine misinformation.
If you have further questions about the COVID-19 vaccine check out the FAQ produced by the Desmond Tutu Health Foundation (DTHF). The DTHF has developed a dedicated chat function where you can ask your vaccine-related questions on the bottom right hand corner of the website.
IDM YouTube channel | IDM website
UCT Community of Hope Vaccination Centre
The University of Cape Town in partnership with the Western Cape Government (WCG) have reinforced our commitment to bringing hope to the residents of the Mother City with the launch of the world‑class Community of Hope Vaccination Centre that opened its doors on Monday, 30 August 2021.
The site is located on Main Road in Mowbray – in the Forest Hill Residence – and access is from Broad Street. The site is open every Monday to Friday from 08:00 to 15:00 and on Saturday from 09:00 to 13:00. Please allow time for attending to COVID-19 protocols and arrive as early as possible at the vaccination centre.
Frequently asked questions
News and views
Campus communications
2021
Media releases
Read more
UCT statements related to COVID-19 vaccinations
This is a space created for all formal bodies and structures within the university community to share their opinions on the need for a mandatory COVID-19 vaccine policy. Please note that some editorial judgement may be applied if the received statements go against any constitutional rights, and that no correspondence will be entered into, statements will be posted unedited and as received. Statements can be sent to opinions@uct.ac.za.
Commemorating a year of COVID-19
At midnight on 26 March 2020, South Africa went into the first nationwide hard lockdown. A year later, we remember those who have died and those who have been affected by COVID-19, as well as the pandemic’s effects across society and campus. We are especially grateful for the front-line health workers who have done so much for so many.
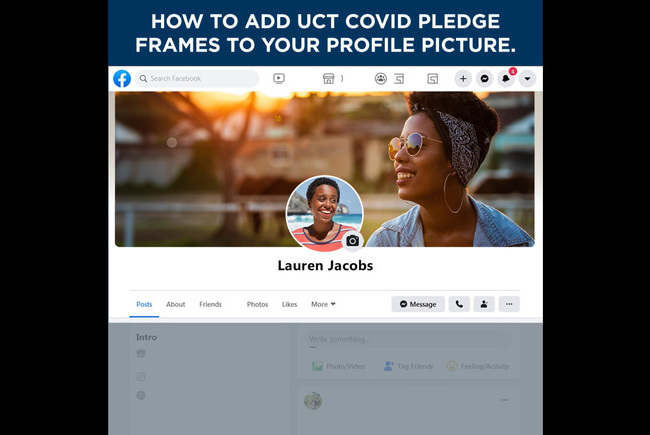
#UCTCOVIDPledge – social media elements
Customised Facebook frames and Instagram stickers are now available on those social media platforms. Watch the tutorial videos here to see how easily you can show your support for the #UCTCOVIDPledge.
In an email to the UCT community, Vice-Chancellor Professor Mamokgethi Phakeng said:
“COVID-19, caused by the virus SARS-CoV-2, is a rapidly changing epidemic. [...] Information [...] will be updated as and when new information becomes available.”
We are continuing to monitor the situation and we will be updating the UCT community regularly – as and when there are further updates. If you are concerned or need more information, students can contact the Student Wellness Service on 021 650 5620 or 021 650 1271 (after hours), while staff can contact 021 650 5685.