Funding boost for low-cost cardiac device
12 December 2022 | Story Nadia Krige. Photo Pixabay Read time 5 min.
A significant funding boost is set to help Strait Access Technology (SAT), a University of Cape Town (UCT) spin-off company, clear the final hurdles in gaining approval for its cost-effective cardiac surgical devices for use in resource-scarce clinical settings.
The funding from Bidvest, which has been supporting SAT since 2012, is set to see the company through until early 2025, says Professor Peter Zilla, SAT CEO and medical director.
Apart from helping to cover the costs of SAT’s 60-plus staff, the funding will hopefully enable it to conduct essential first-in-human trials.
“We are optimistic that within this funding cycle, we will have concluded key clinical trials that will make us attractive for a partnership with a major medical device company,” he says.
A cost-effective intervention
Founded in 2008, SAT’s objective is to develop intelligent surgical products for treating rheumatic heart disease in emerging economies with a strong focus on Africa, which is considered one of the epicentres of the condition.
Rheumatic heart disease causes permanent damage to heart valves and typically develops as a delayed side effect of untreated or under-treated infection with the bacteria Streptococcus, also known as strep. This is mostly due to a lack of access to primary healthcare and medication to treat the infection, putting people living in rural communities most at risk.
There are no transcatheter devices for rheumatic heart disease.
Currently, the only way to replace heart valves for rheumatic patients is with open-heart surgery, which requires opening their chest, stopping their heart, cutting out the diseased valve and sewing in a replacement.
In addition to being invasive and costly, this surgery can be done at only 48 centres in South Africa, of which 14 are in Cape Town. And of those 14 in Cape Town, most are for the small part of the population (13%) with medical aid.
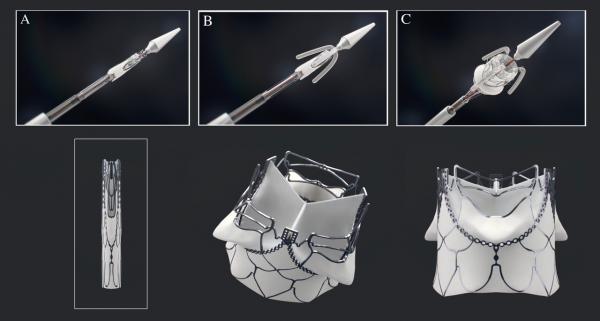
The main product the SAT team is developing is a transcatheter aortic valve implantation (TAVI) system that allows clinicians to perform valve-replacement surgery in a fraction of the time open-heart surgery takes and for a fraction of the costs.
“At the moment, there are no transcatheter devices for rheumatic heart disease, and those that exist in clinical use are all for typical western patients with narrowed valves,” says Zilla. “Rheumatic heart disease, most common among the indigent population, is typically associated with leaking heart valves, and for that, there is no transcatheter device.”
With the TAVI system, surgeons can make a tiny incision of about a centimetre and deploy the new valve using an insertion device and a balloon-expandible stent. The procedure is guided mainly by tactile feedback supported by sonar imaging, which also makes it more applicable to rural settings.
Overcoming obstacles
Having proven itself to be a well-established university start-up, the company was making promising strides towards gaining approval for the use of its devices in clinical settings when COVID-19 hit South African shores in early 2020.
“We were really seriously affected by the pandemic,” says Zilla. “You can’t remote-produce and remote-test devices, and we were just in that phase where we needed to be doing both.”
This was followed by another major blow as SAT lost seven of its senior engineers to European companies within 15 months. This brain drain resulted from a drive to recertify products after the European Union revised its medical device regulations in May 2021.
“Fortunately, we have refilled all the positions now,” Zilla says. “We are also proud that 80% of these are now filled by outstanding female engineers.”
Clinical trials on the cards
The final obstacle Zilla and his team face is amending SAT’s approval from the South African Health Products Regulatory Authority (SAHPRA) to conduct its first-in-human trials. The initial inclusion criteria for patients to participate were too narrowly defined by the university’s ethics committee.
We were really seriously affected by the pandemic. You can’t remote-produce and remote-test devices.
The problem came down to the conflict of conducting trials for medical devices designed for low-resource settings in comparatively resource-rich environments, such as Groote Schuur Hospital.
“We have one more meeting this year with the ethics committee,” says Zilla. “I am rather optimistic that we have answered them so conscientiously and thoroughly that we could possibly look forward to recruiting our first patients in early 2023.”
Should this be the case, SAT will start with a safety cohort of about 10 patients to prove that the device does no harm, after which they will apply for approval to recruit a larger cohort of patients for further trials.
The funding SAT received from Bidvest will play a crucial role in supporting it through these next steps.
 This work is licensed under a Creative Commons Attribution-NoDerivatives 4.0 International License.
This work is licensed under a Creative Commons Attribution-NoDerivatives 4.0 International License.
Please view the republishing articles page for more information.
Research & innovation





































